- $21,124,760All Federal Fiscal Year 2022 Research Funding14thOut of 142 in 2022 research dollars to colleges of pharmacy
Overview
The College of Pharmacy’s extensive research is funded by over $15 million in extramural grants, research awards and industry partnerships. The College is ranked 10th out of 142 in 2018 NIH research dollars to colleges of pharmacy. Our state of the art laboratories and support clusters in Avedisian Hall are home to 35 faculty researchers and their research teams.
Focus Areas
Faculty in the URI College of Pharmacy are respected experts in the biomedical, clinical and applied pharmaceutical sciences. Their primary areas of research, interest, and expertise are outlined below. A searchable database of health and research expertise can be found at the Health Expert Database.
Drug Discovery
Where will we find the next generation of antibiotics? Can natural products augment or replace our current therapies for cancer and dementia? Many of humankind’s most devastating diseases have few or no effective therapies. Researchers in the College of Pharmacy are using a variety of approaches to identify and develop new strategies for treating antibiotic-resistant infections, cancer and neurodegenerative diseases. These investigators are collaborating with local and federal agencies and industry partners to translate laboratory results into real-world solutions.
Drug Delivery
Sustained release technologies, transporters, nanoparticles, powders and aerosols are among the drug delivery strategies being investigated today at the College of Pharmacy. Researchers are developing new approaches for delivering therapies across physiological barriers to enhance the treatment of a wide variety of diseases.
Pharmaceutical Chemistry
The development of new drugs and drug delivery strategies is dependent on a solid understanding of the chemistry of the medication and its vehicle. Pharmacy faculty employ a number of sophisticated technologies to probe the chemistry of organic and inorganic molecules, including Mass Spectrometry, NMR Spectroscopy, HPLC and nucleic acid synthesis and isolation. In many cases these investigators share their expertise and capabilities with pharmaceutical developers and manufacturers (large and small) in relationships that benefit the College as well as our industry partners.
Pharmacology
Pharmacologists in the College of Pharmacy possess expertise in a variety of sub-disciplines, including clinical pharmacology, drug metabolism, pharmacokinetics, pharmacodynamics, clinical study design and the analysis of drug response within populations. In many cases, these studies are designed to develop an understanding the sources of variability in drug disposition and effect and in the identification of individual factors that lead to altered drug response.
Toxicology
A number of the investigators in the College of Pharmacy study the consequences of exposure to environmental toxins. These investigators have particular interests in the relationships between the environment and Alzheimer’s disease, fatty liver disease, obesity and kidney disease. Their work has led to significant advancements in our understanding of the environment’s impact on the epigenome (factors that influence gene expression without changing the genetic code).
Pharmacotherapy
A large proportion of the College of Pharmacy faculty conduct research related to the practice of pharmacy. In some cases this work focuses on the treatment of a specific disease, such as diabetes, while in other instances the research involves the study of populations, e.g. the elderly. Many COP faculty members have a strong interest in developing practices that will improve the dissemination of drug-related information and enhance patient compliance.
Clinical Study Design and Analysis
The value of clinical studies is directly related to the quality of the study design and its subsequent analysis. A number of Pharmacy faculty members are recognized experts in this field. These investigators participate in a variety of partnerships and collaborations at the local, regional and national level.
Health Policy and Health Outcomes
The rapidly evolving nature of the healthcare marketplace in the United States requires that health-related decisions be appropriately informed by sound health policy and accurate economic analyses. Pharmacy faculty provide consulting and research support to government agencies and a variety of healthcare organizations and help to develop strategies that maximize the quality of care, improve strategic and operational efficiency and optimize the allocation of resources.
News
 Pharmacies should distribute naloxone along with opioids, URI College of Pharmacy study shows - Medication can quickly stop an opioid-related overdose, saving lives By Allie Shinskey Pharmacies across the region are being encouraged to distribute naloxone along with higher dose opioid prescriptions and medication combinations to help fight the opioid overdose crisis, according to a recent University of Rhode Island College of Pharmacy study. Naloxone, a safe and effective […]
Pharmacies should distribute naloxone along with opioids, URI College of Pharmacy study shows - Medication can quickly stop an opioid-related overdose, saving lives By Allie Shinskey Pharmacies across the region are being encouraged to distribute naloxone along with higher dose opioid prescriptions and medication combinations to help fight the opioid overdose crisis, according to a recent University of Rhode Island College of Pharmacy study. Naloxone, a safe and effective […] URI, BayCare in Florida and Butler Hospital team up to test retinal scanning for early detection of Alzheimer’s disease - University of Rhode Island College of Pharmacy professors are part of a clinical trial of retinal screening processes that could help clinicians detect Alzheimer’s disease possibly two or more decades before patients develop life-altering clinical symptoms.
URI, BayCare in Florida and Butler Hospital team up to test retinal scanning for early detection of Alzheimer’s disease - University of Rhode Island College of Pharmacy professors are part of a clinical trial of retinal screening processes that could help clinicians detect Alzheimer’s disease possibly two or more decades before patients develop life-altering clinical symptoms. Toxic algal blooms may be key to slowing neurodegenerative disease - Pharmacy Professor Matthew Bertin teams with pharmaceutical firm to discover anti-inflammatory molecules from aquatic microbiome Toxic algal blooms can be devastating to natural waterways, robbing them of oxygen, creating dead zones, and sickening people and animals. However, they may also be beneficial, potentially helping combat the progression of neurodegenerative disease, a URI College of Pharmacy […]
Toxic algal blooms may be key to slowing neurodegenerative disease - Pharmacy Professor Matthew Bertin teams with pharmaceutical firm to discover anti-inflammatory molecules from aquatic microbiome Toxic algal blooms can be devastating to natural waterways, robbing them of oxygen, creating dead zones, and sickening people and animals. However, they may also be beneficial, potentially helping combat the progression of neurodegenerative disease, a URI College of Pharmacy […] URI, JWU and FIX combine to unlock power of ‘The Smarter Sweetener’ - Culinary, design students apply health benefits research uncovered in maple Students from around the region created tasty treats and demonstrated innovative uses of a local crop that shows promising health benefits as they demonstrated the culinary — and medicinal — versatility of maple. The Maple internship program at the Food Innovation Nexus (FIX) at Johnson […]
URI, JWU and FIX combine to unlock power of ‘The Smarter Sweetener’ - Culinary, design students apply health benefits research uncovered in maple Students from around the region created tasty treats and demonstrated innovative uses of a local crop that shows promising health benefits as they demonstrated the culinary — and medicinal — versatility of maple. The Maple internship program at the Food Innovation Nexus (FIX) at Johnson […] URI awarded $20 million to further expand biomedical research capacity in R.I. - A University of Rhode Island-based initiative that has successfully expanded biomedical research capacity at nearly all of Rhode Island’s universities and colleges has been awarded another $20 million in federal funding to further expand the program over the next five years.
URI awarded $20 million to further expand biomedical research capacity in R.I. - A University of Rhode Island-based initiative that has successfully expanded biomedical research capacity at nearly all of Rhode Island’s universities and colleges has been awarded another $20 million in federal funding to further expand the program over the next five years. URI College of Pharmacy ranked 10th nationally in federal research funding - The College attracted more than $12 million in federal funds in fiscal 2018 The University of Rhode Island College of Pharmacy has moved into the top 10 in the country in total federal research grant funding from the National Institutes of Health after securing more than $12 million in federal research funds in fiscal 2018. […]
URI College of Pharmacy ranked 10th nationally in federal research funding - The College attracted more than $12 million in federal funds in fiscal 2018 The University of Rhode Island College of Pharmacy has moved into the top 10 in the country in total federal research grant funding from the National Institutes of Health after securing more than $12 million in federal research funds in fiscal 2018. […] URI College of Pharmacy study to examine whether opioid prescription rates impact a community’s overall health - By Ross Balding Is there a link between high opioid prescription rates and mortality? Does an increase in opioid prescriptions worsen a community’s overall health expectancy? What role do health care providers play in the ever-worsening opioid crisis in the country? University of Rhode Island College of Pharmacy Professors Ashley Buchanan and Jeffrey Bratberg aim […]
URI College of Pharmacy study to examine whether opioid prescription rates impact a community’s overall health - By Ross Balding Is there a link between high opioid prescription rates and mortality? Does an increase in opioid prescriptions worsen a community’s overall health expectancy? What role do health care providers play in the ever-worsening opioid crisis in the country? University of Rhode Island College of Pharmacy Professors Ashley Buchanan and Jeffrey Bratberg aim […] Ryan Institute Gets Approval for Groundbreaking Alzheimer’s Clinical Trial - College of Pharmacy’s Pharmaceutical Development Institute plays key role in trial.
Ryan Institute Gets Approval for Groundbreaking Alzheimer’s Clinical Trial - College of Pharmacy’s Pharmaceutical Development Institute plays key role in trial. Fall Color In-Depth: Maple Trees Offer New Answers to Diabetes, Alzheimer’s - In the 1992 film Medicine Man, biochemist Robert Campbell, played by actor Sean Connery, searches for new drugs in the Amazon’s vast rainforests. There Campbell finds a cure for cancer not in the rainforest’s rare flowers – which don’t have “juju,” or the power to heal – but in an indigenous ant species.
Fall Color In-Depth: Maple Trees Offer New Answers to Diabetes, Alzheimer’s - In the 1992 film Medicine Man, biochemist Robert Campbell, played by actor Sean Connery, searches for new drugs in the Amazon’s vast rainforests. There Campbell finds a cure for cancer not in the rainforest’s rare flowers – which don’t have “juju,” or the power to heal – but in an indigenous ant species. Private Support Advances Research on Medicinal Properties of Maple Trees - While the maple tree is known for delicious syrup, researchers at URI have discovered that maple trees provide much more than a way to make breakfast taste better. In its branches, leaves, and sap, maple species, including Rhode Island’s state tree, the red maple, may be the key to regulating blood glucose levels in humans. Thanks to $110,000 in funding from Verdure Sciences, this important research will continue.
Private Support Advances Research on Medicinal Properties of Maple Trees - While the maple tree is known for delicious syrup, researchers at URI have discovered that maple trees provide much more than a way to make breakfast taste better. In its branches, leaves, and sap, maple species, including Rhode Island’s state tree, the red maple, may be the key to regulating blood glucose levels in humans. Thanks to $110,000 in funding from Verdure Sciences, this important research will continue.
Facilities
 Heber W. Youngken Jr. Medicinal Garden - An educational display of medicinal plants herbs and spices and also as a conservatory source of standard specimens.
Heber W. Youngken Jr. Medicinal Garden - An educational display of medicinal plants herbs and spices and also as a conservatory source of standard specimens. Pharmaceutical Development Institute (PDI) - The PDI provides contract development and testing services for solid oral dosage forms specializing in early development. In addition, the PDI offers training workshops for pharmaceutical and biopharmaceutical companies. The workshops offer classroom instruction and hands-on training that can be custom-tailored to the company’s unique needs.
Pharmaceutical Development Institute (PDI) - The PDI provides contract development and testing services for solid oral dosage forms specializing in early development. In addition, the PDI offers training workshops for pharmaceutical and biopharmaceutical companies. The workshops offer classroom instruction and hands-on training that can be custom-tailored to the company’s unique needs. RI-INBRE Centralized Research Core Facility - A staffed core instrumentation facility supporting research and training for RI-INBRE participants and the wider Rhode Island biomedical research community.
RI-INBRE Centralized Research Core Facility - A staffed core instrumentation facility supporting research and training for RI-INBRE participants and the wider Rhode Island biomedical research community.
Labs & Projects
 Advanced Drug Delivery & Bioengineering Research Laboratory (Shen) - The Shen Lab is specialized in the areas of formulation development, manufacturing, in vitro and in vivo product performance testing, and development of in vitro-in vivo correlation (IVIVC) for advanced drug delivery systems and medical devices.
Advanced Drug Delivery & Bioengineering Research Laboratory (Shen) - The Shen Lab is specialized in the areas of formulation development, manufacturing, in vitro and in vivo product performance testing, and development of in vitro-in vivo correlation (IVIVC) for advanced drug delivery systems and medical devices. Antimicrobial Stewardship Program (Laplante) - A collaborative research program within the University of Rhode Island College of Pharmacy, the Antimicrobial Stewardship Program focuses on the prevention, treatment, virulence inhibition, and outcomes associated with drug resistant bacteria.
Antimicrobial Stewardship Program (Laplante) - A collaborative research program within the University of Rhode Island College of Pharmacy, the Antimicrobial Stewardship Program focuses on the prevention, treatment, virulence inhibition, and outcomes associated with drug resistant bacteria. Bioactive Botanical Laboratory (Seeram) - Inside URI’s Bioactive Botanical Research Lab, Pharmacy Professor Navindra Seeram has some big ideas about fighting cancer. Ideas so big that he’s regularly sought after by science and news publications around the globe for his expertise in the healing properties of medicinal plants.
Bioactive Botanical Laboratory (Seeram) - Inside URI’s Bioactive Botanical Research Lab, Pharmacy Professor Navindra Seeram has some big ideas about fighting cancer. Ideas so big that he’s regularly sought after by science and news publications around the globe for his expertise in the healing properties of medicinal plants. Chemical Carcinogenesis Research (Cho) - Our research area is in chemical carcinogenesis with a major emphasis on the structural and synthetic aspects of DNA-adduct formation. Aromatic amine and polycyclic aromatic hydrocarbons are present in environment, thereby posing a threat to human health. I am particularly interested in how environmental carcinogens, upon metabolism, interact with cellular DNA and initiate mutagenesis/carcinogenesis. The […]
Chemical Carcinogenesis Research (Cho) - Our research area is in chemical carcinogenesis with a major emphasis on the structural and synthetic aspects of DNA-adduct formation. Aromatic amine and polycyclic aromatic hydrocarbons are present in environment, thereby posing a threat to human health. I am particularly interested in how environmental carcinogens, upon metabolism, interact with cellular DNA and initiate mutagenesis/carcinogenesis. The […]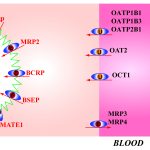 Clinical Pharmacokinetics and Pharmacodynamics (Rosenbaum) - Dr. Rosenbaum is applying modeling and simulation of drug response data as tools to more fully understand the time course and intensity of drug effects in man. She is interested in applying these methods in research to obtain a greater understanding of a drug’s properties in different populations.
Clinical Pharmacokinetics and Pharmacodynamics (Rosenbaum) - Dr. Rosenbaum is applying modeling and simulation of drug response data as tools to more fully understand the time course and intensity of drug effects in man. She is interested in applying these methods in research to obtain a greater understanding of a drug’s properties in different populations. Deng Lab - Dr. Deng’s research program is primarily focused on investigating bile acid and cholesterol homeostasis under physiological and pathological conditions. Disruption of bile acids and cholesterol homeostasis by genetic or environmental factors has been associated with various diseases.
Deng Lab - Dr. Deng’s research program is primarily focused on investigating bile acid and cholesterol homeostasis under physiological and pathological conditions. Disruption of bile acids and cholesterol homeostasis by genetic or environmental factors has been associated with various diseases. Maple Collaborative - Providing information to the maple benefits to promote the maple industry.
Maple Collaborative - Providing information to the maple benefits to promote the maple industry. Nanotherapeutics & Biomaterials Engineering Laboratory (Menon) - The Dr. Menon lab’s research is centered on using drug delivery- and tissue engineering – based approaches to develop novel and effective solutions to cancer, fibrosis and inflammation. Our current research focuses are
Nanotherapeutics & Biomaterials Engineering Laboratory (Menon) - The Dr. Menon lab’s research is centered on using drug delivery- and tissue engineering – based approaches to develop novel and effective solutions to cancer, fibrosis and inflammation. Our current research focuses are Natural Product Drug Discovery Research (Rowley) - One of the more frightening medical trends today is our growing resistance to antibiotics, which we take for everything from ear infections to life-threatening blood conditions. As people use antibiotics for more and more ailments, the ability of germs and bacteria to mutate and adapt has increased, often making them stronger than the medicines available […]
Natural Product Drug Discovery Research (Rowley) - One of the more frightening medical trends today is our growing resistance to antibiotics, which we take for everything from ear infections to life-threatening blood conditions. As people use antibiotics for more and more ailments, the ability of germs and bacteria to mutate and adapt has increased, often making them stronger than the medicines available […] Rhode Island Infectious Diseases Research Program - Antimicrobial resistance is globally recognized as one of the greatest healthcare threats. Our research program is focused on the prevention, treatment, virulence inhibition and clinical outcomes associated with drug resistant bacteria. The program is divided into three divisions: the antimicrobial pharmacology division, the outcomes and pharmacoepidemiology division, and the implementation science outreach division.
Rhode Island Infectious Diseases Research Program - Antimicrobial resistance is globally recognized as one of the greatest healthcare threats. Our research program is focused on the prevention, treatment, virulence inhibition and clinical outcomes associated with drug resistant bacteria. The program is divided into three divisions: the antimicrobial pharmacology division, the outcomes and pharmacoepidemiology division, and the implementation science outreach division.
